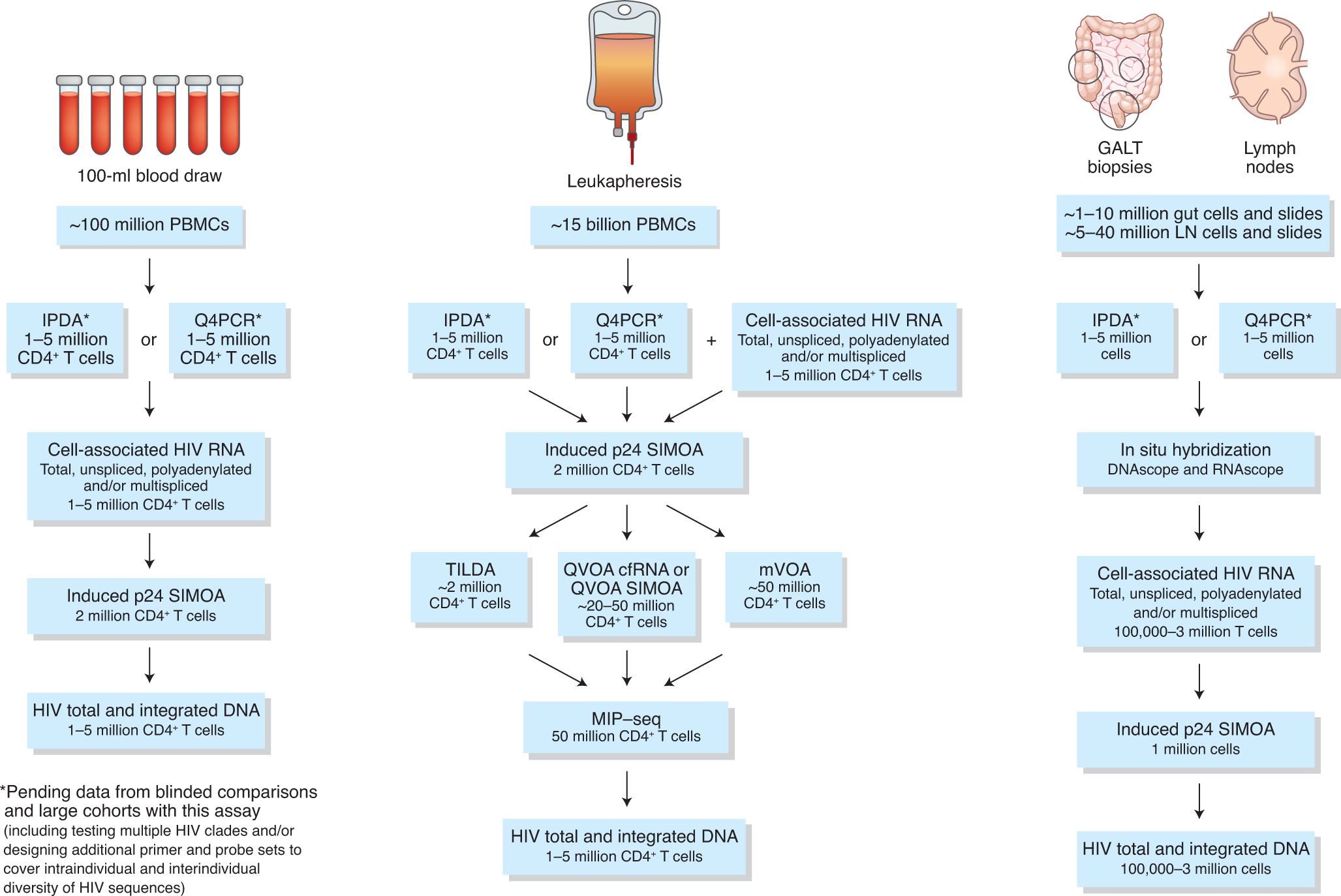
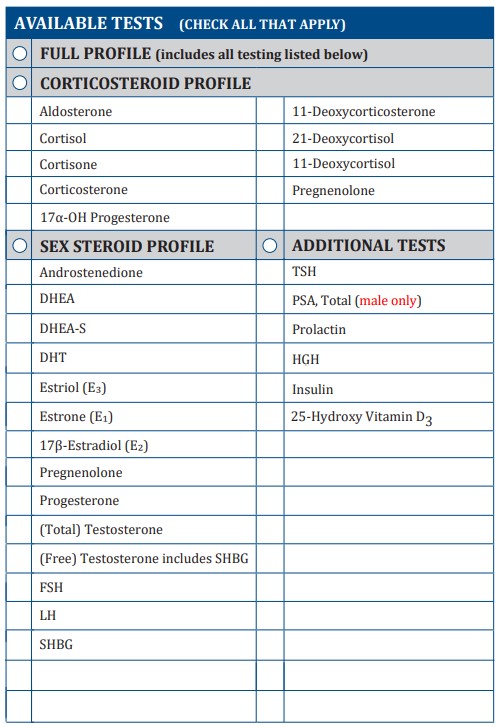
On Biostatistics and Clinical Trials: Guidelines for Blood Volumes in Clinical Trials (Especially in Pediatric Clinical Trials)
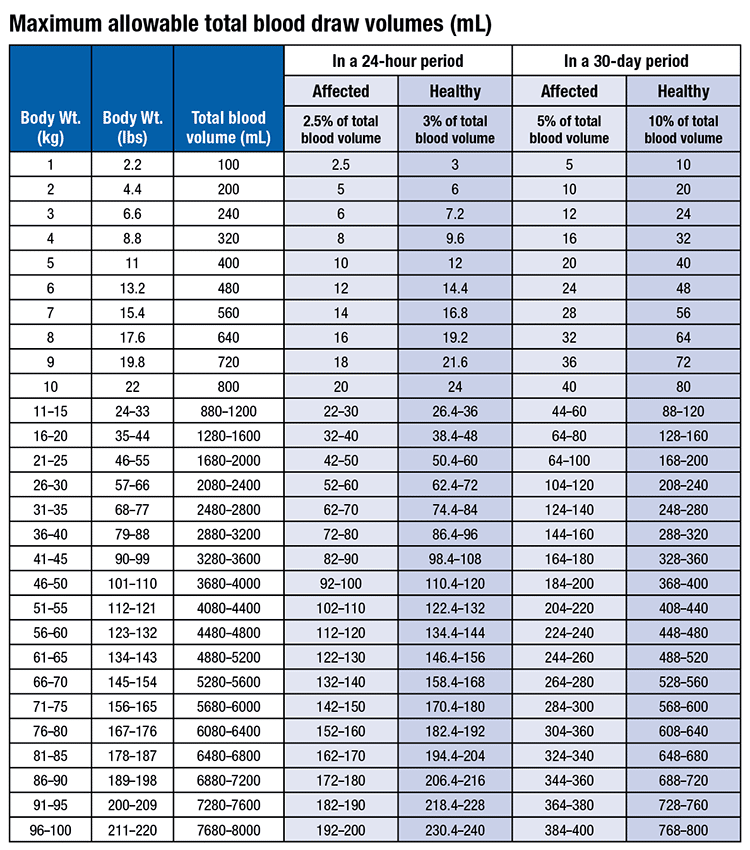
ACCEPTABLE BLOOD VOLUMES DRAWN FOR CHILREN IN RESEARCH STUDIES CHLA IRB Blood Draw Volume Policy CHLA IRB policy is to limit th
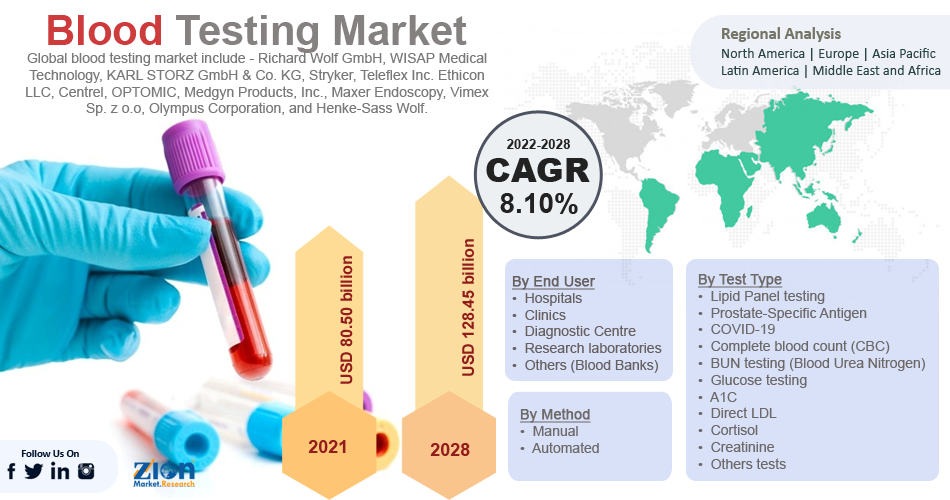
Blood Testing Market Report by Size, Analysis, Future Growth, Global Trends, and Forecast 2028 | Grow at CAGR of 8.10 %